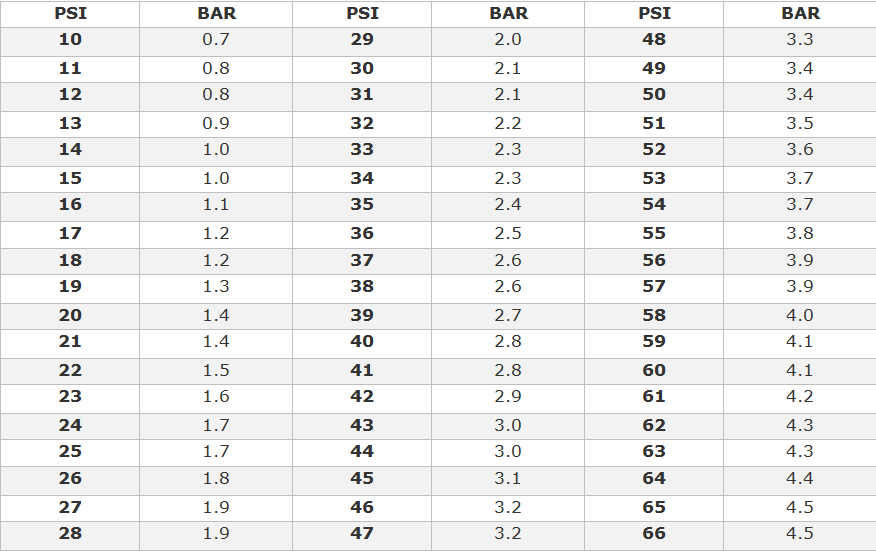
Therefore, this table uses "torr" as both the name and the symbol.Ī pressure of 1 atm can also be stated as: National Institute of Standards and Technology found no such clear-cut definitions. An extensive search of the website of the U.S. Both the United Kingdom's National Physical Laboratory (see Pressure Units) and New Zealand's Measurement Standards Laboratory (see Barometric Pressure Units) use "torr" as the name and as the symbol. Nor is there any consensus about whether the symbol for that unit of pressure should be "Torr" or "torr". Note: mmHg is an abbreviation for millimetre of mercury About the torr: There is no consensus in the technical literature about whether the name of the torr should be "Torr" or "torr". Pressure units and equivalents Pressure UnitsĮxample reading: 1 Pa = 1 N/m 2 = 10 −5 bar = 9.8692×10 −6 atm = 7.5006×10 −3 torr, etc. However, the atm continues to be used quite often as a unit of pressure. However, in 1982, the International Union of Pure and Applied Chemistry (IUPAC) recommended that for the purposes of specifying the physical properties of substances, the "standard pressure" should be defined as precisely 100 kPa (exactly 1 bar). In chemistry, the original definition of "Standard Temperature and Pressure" was a reference temperature of 0 ☌ (273.15 K) and pressure of 101.325 kPa (1 atm). This value was intended to represent the mean atmospheric pressure at mean sea level at the latitude of Paris, France, and as a practical matter, truly reflects the mean sea level pressure for many of the industrialized nations (those with latitudes similar to Paris). In 1954 the 10th Conférence Générale des Poids et Mesures (CGPM) adopted standard atmosphere for general use and affirmed its definition of being precisely equal to 1,013,250 dynes per square centimeter (101,325 Pa). The difference between an atm and a bar, which is about 1%, is not significant for many applications, and is within the error range of common pressure gauges. For practical purposes, it is often replaced by the bar, defined as 100,000 Pa. 
An atmosphere (symbol: atm) is a unit of pressure measurement defined as 101,325 Pa.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
